 Now you can find answers to all your subject queries & prepare for your Exams on our Learning App – Infinity Learn. 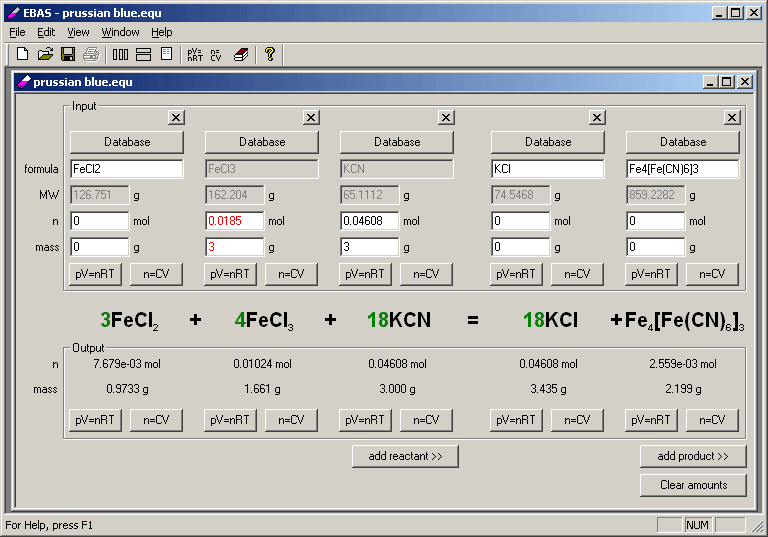 
Volume of Mohr’s salt solution taken for each titration = 20.0 mlįrom the overall balanced chemical equation, it is clear that 2 moles of KMnO 4 reacts with 10 moles of Mohr’s salt. Volume of Mohr’s salt solution prepared = 250 ml Aluminum Chloride + Sulfuric Acid Aluminum Sulfate + Hydrogen Chloride. Lithium Hydroxide + Sulfuric Acid Lithium Sulfate + Water. Aluminum Hydroxide + Sulfuric Acid Aluminum Sulfate + Water. Weight of watch glass + Mohr’s salt =………….g
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
